Neuroform Atlas™ Stent System

Bending expectations of conformability and stability The Neuroform Atlas Stent System is authorized under a Humanitarian Device Exemption (HDE). IRB approval is required prior to use.
Learn moreNeuroform EZ® Stent System

Flexible design. Enhanced delivery. The Neuroform EZ Stent System is authorized under a Humanitarian Device Exemption (HDE). IRB approval is required prior to use.*
Learn moreTarget XL® Detachable Coils

Larger and longer Target XL Coils provide two times more fill and 40% more width than Target 10-size coils. All deliverable through Excelsior® SL-10® and XT-17™ Microcatheters.
Learn moreTarget® 360 Detachable Coils

Conformability from frame to finish Open loop configuration designed to conform to different aneurysm shapes, fill uniformly, and minimize compartmentalization.
Learn moreTarget® 3D Detachable Coils
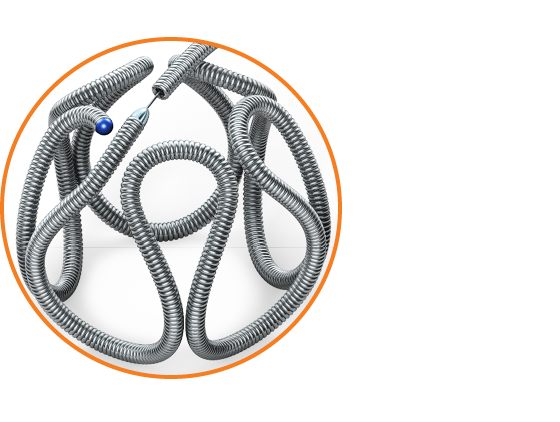
Consistent framing Alternating large and small loops are designed to create a stable frame.
Learn moreTarget® Nano™ Detachable Coils

Beyond soft 2x softer than Target Ultra Coils with advanced microcatheter stability. Designed to fill small spaces and conform to the final spaces within aneurysms.
Learn moreTransForm® Occlusion Balloon Catheter

Fast. Easy. Stable. The TransForm Occlusion Balloon Catheter is a 0.014in guidewire compatible, single lumen occlusion balloon designed to provide enhanced performance.
Learn more- * The Neuroform® Microdelivery Stent System is authorized by Federal Law for use with embolic coils for the treatment of wide-neck, intracranial, saccular aneurysms arising from a parent vessel with a diameter of ≥ 2mm and ≤ 4.5mm that are not amenable to treatment with surgical clipping. Wide-neck aneurysms are defined as having a neck ≥ 4mm or a dome-to-neck ratio < 2. The effectiveness of this device for this use has not been demonstrated. IRB Approval Required. Please contact your local sales representative for details about ordering this product.